In the VICTORIA trial, VERQUVO demonstrated an adverse event profile similar to placebo
VERQUVO is well tolerated by patients following a HF hospitalization or use of outpatient IV diuretics
The adverse drug reactions occurring more commonly with VERQUVO than placebo and in ≥5% of patients treated with VERQUVO in VICTORIA

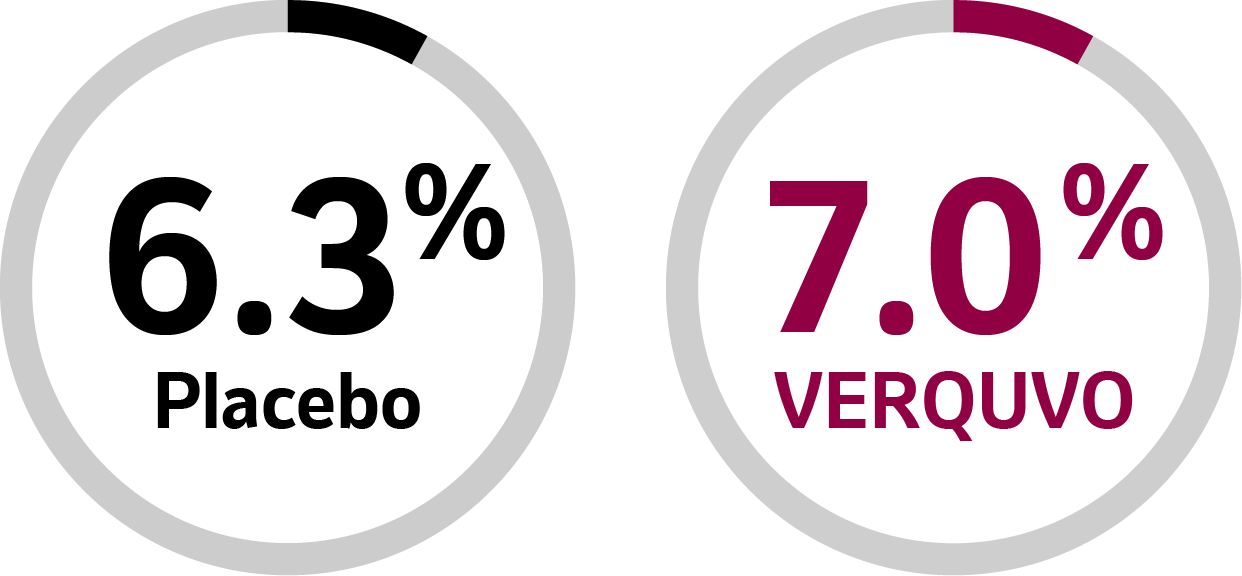
7.0% of patients taking VERQUVO discontinued due to AEs1
AEs = adverse events.
Treatment initiation with VERQUVO 5 mg once daily in VELOCITY was similarly tolerated as treatment initiation of 2.5 mg once daily in the VICTORIA study.
aBackground therapy included beta blocker, ACE inhibitor, ARB, MRA, ARNI, and/or SGLT2 inhibitor.
Study designs
VICTORIA was a Phase 3, randomized, parallel-group, placebo-controlled, double-blind, event-driven, multicenter trial comparing VERQUVO to placebo when added to background HF therapy in 5,050 adult patients with NYHA class II-IV chronic HF and LVEF <45% following a worsening HF event (defined as HF hospitalization within 6 months before randomization or use of outpatient IV diuretics for HF within 3 months before randomization). Patients were treated up to the target maintenance dose of VERQUVO 10 mg once daily or matching placebo. The primary endpoint was a composite of time to first event of CV death or HF hospitalization.1
VELOCITY was a Phase 2b, 2-week, open-label, single-arm, multinational prospective trial evaluated the safety and tolerability of VERQUVO 5 mg once daily as a starting dose. The trial enrolled 106 adult patients aged ≥18 years with chronic heart failure and a left ventricular ejection fraction <45%, New York Heart Association class II–IV symptoms, and systolic blood pressure ≥100mmHg at screening and Visit 1. Study participants with or without worsening heart failure on background guideline-directed medical therapy for heart failure underwent screening for eligibility. Eligible patients returned 2 weeks later for Visit 1. At this Visit 1, VERQUVO 5 mg daily was initiated and continued for 2 weeks as tolerated until the final visit (Visit 2). The primary endpoint was to assess the tolerability of a 5 mg VERQUVO starting dose, defined as completing the 2-week course with no more than a 1-day interruption and without moderate-to-severe symptomatic hypotension.2
Definitions:
ACE = angiotensin-converting enzyme; AEs = adverse events; ARB = angiotensin II receptor blocker; ARNI = combination of an angiotensin receptor and neprilysin inhibitor; CV = cardiovascular; HF = heart failure; IV = intravenous; LVEF = left ventricular ejection fraction; MRA = mineralocorticoid receptor antagonist; NYHA = New York Heart Association; SGLT2 = sodium glucose co-transporter 2; VICTORIA = Vericiguat in Patients with Heart Failure and Reduced Ejection Fraction.
Reference:
- Armstrong PW, Pieske B, Anstrom KJ, et al. Vericiguat in patients with heart failure and reduced ejection fraction. N Engl J Med. 2020;382(20):1883-1893.
- Greene SJ, Corda S, McMullan CJ, et al, on behalf of the VELOCITY investigators. Safety and tolerability of a 5 mg starting dose of vericiguat among patients with heart failure: The VELOCITY study. Eur J Heart Fail. 2025(27):2837-2840. https://doi.org/10.1002/ejhf.3699
