Study designs in adult hematopoietic stem cell transplantation (HSCT) patients
Prophylaxis through Week 14 (~100 days) post-HSCT in adults
Prophylaxis from Week 14 (~100 days) through Week 28 (~200 days) post-HSCT in adults
Pediatric information for HSCT
Prophylaxis through Week 14 (~100 days) post-HSCT in adults
Cytomegalovirus (CMV) prophylaxis was assessed in a randomized, multicenter, double-blind, placebo-controlled, pivotal, phase 3 study of adult CMV-seropositive recipients [R+] of allogeneic hematopoietic stem cell transplant (HSCT). Patients were randomized 2:1 to PREVYMIS or placebo and stratified by study site and high vs low risk (N=565).
- All-cause mortality through Week 48 post-transplant was a prespecified, exploratory analysis.1
- The protocol-specified guidance for CMV DNA thresholds for the initiation of preemptive therapy (PET) during the treatment period were ≥150 copies/mL or >300 copies/mL for subjects in the high- and low-risk strata, respectively.
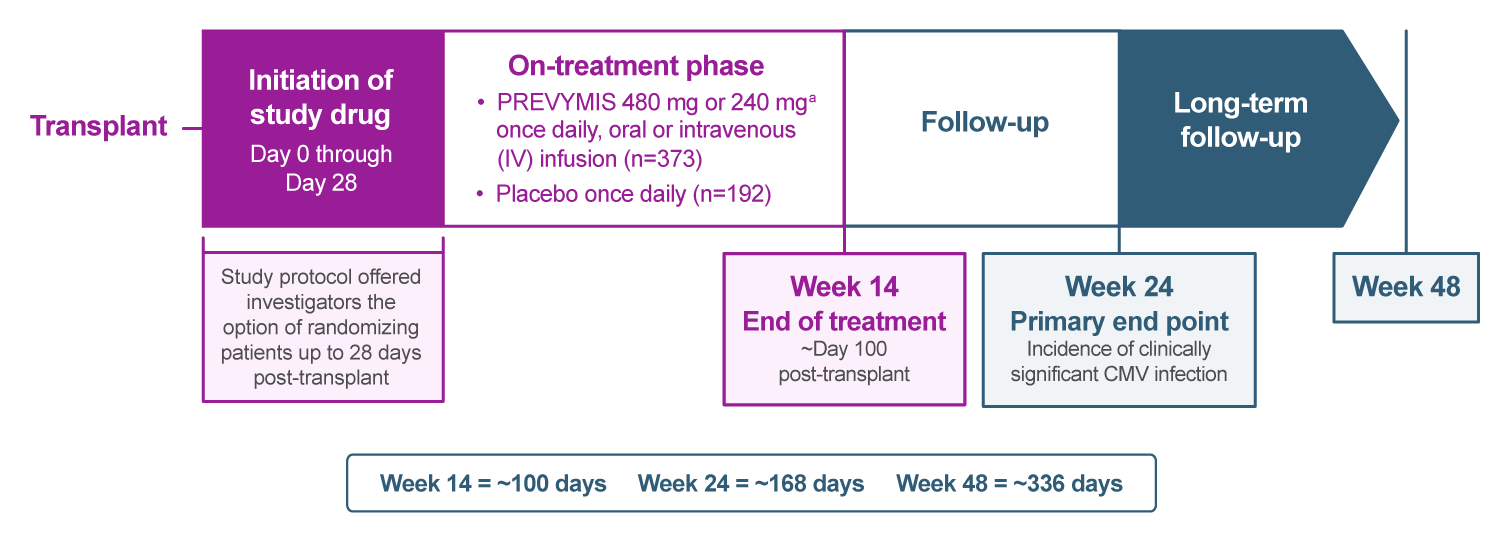
(a) When coadministered with cyclosporine, the dose of PREVYMIS was decreased to 240 mg once daily, oral or IV infusion.
- Patients were considered at increased risk for CMV reactivation if they had 1 or more of the following factors:
- Human leukocyte antigen (HLA)-related donor with at least 1 mismatch at 1 of 3 HLA-gene loci: HLA-A, -B, or -DR
- Haploidentical donor; unrelated donor with at least 1 mismatch at 1 of 4 HLA-gene loci: HLA-A, -B, -C, or -DRB1
- Use of umbilical cord blood as the stem cell source
- Use of ex vivo T-cell–depleted grafts
- Grade 2 or greater graft-versus-host disease (GVHD), requiring systemic corticosteroids
Prophylaxis from Week 14 (~100 days) through Week 28 (~200 days) post-HSCT in adults
The efficacy of extending PREVYMIS prophylaxis from Week 14 (~100 days) through Week 28 (~200 days) post-HSCT in adult patients at risk for late CMV infection and disease was assessed in a multicenter, double-blind, placebo-controlled Phase 3 trial in adult CMV-seropositive recipients [R+] of an allogeneic HSCT. Eligible subjects who completed PREVYMIS prophylaxis through ~100 days post-HSCT were randomized (2:1) to receive PREVYMIS or placebo from Week 14 through Week 28 post-HSCT.
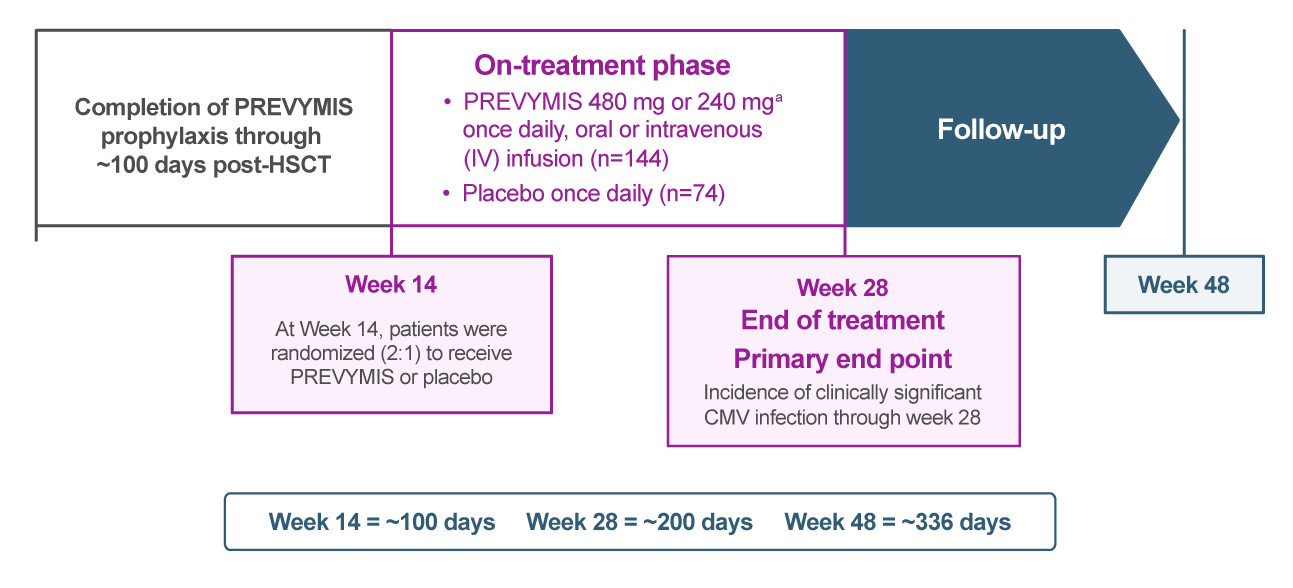
(a) When coadministered with cyclosporine, the dose of PREVYMIS was adjusted to 240 mg once daily, oral or IV infusion.
Reference
- Marty FM, Ljungman P, Chemaly RF, et al. Letermovir prophylaxis for cytomegalovirus in hematopoietic-cell transplantation. N Engl J Med. 2017;377(25):2433–2444. doi:10.1056/NEJMoa1706640